Solid krypton element1/1/2024 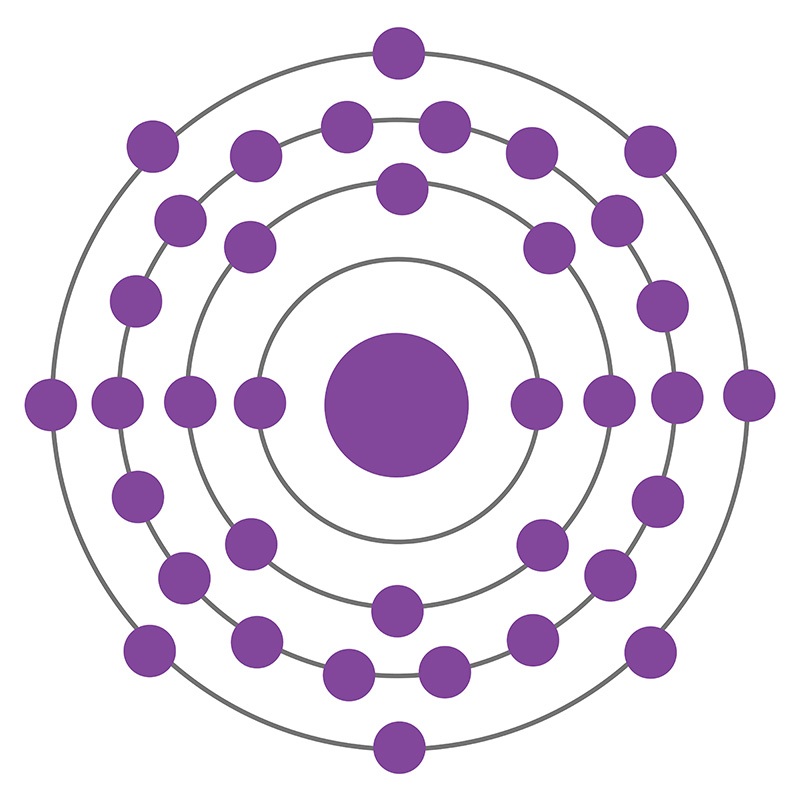
We hope you enjoyed this month’s featured element: Krypton. Krypton LED bulbs shine with an intense white light that makes it incredibly useful for high-speed cameras like those used by our friends at Questacon in the videos below. You can also find it in really expensive lights for photography. When an electrical current is passed through a tube filled with krypton, all of the atoms of the gas get super excited and emit blue light. If you’ve ever seen “neon” lights, not all of the gas inside of those brightly-coloured tubes is what it says on the tin, so to speak. Krypton is also used in fluorescent lighting. Nowadays, we use the distance travelled by a light in 1/299,792,458th of a second – apparently, it’s a bit easier to measure. Between 19, we used one of these lines (multiplied by 1,650,763.73!) to define the metre for every country in the world that used it. Scientists use them to figure out what’s in stars in the galaxy as well as materials here on Earth. It’s basically a unique set of lines produced when you shine light onto an element. Krypton has a melting point of -157.4oC and a. Krypton in action…įor a little while, Krypton was the basis for the definition of the metre! Every element has what’s called an “emission spectrum”. Krypton has a face-centred cubic crystalline structure in its solid state, similar to other noble gas elements. It reacts with fluorine to form a special type of laser that engineers use to make tiny computer chips. In fact, the only thing Krypton seems remotely interested in is the gas fluorine. As we mentioned before, as a noble gas it doesn’t react with very much at all. Krypton does have a few interesting uses here on our planet. Since it only really exists in our atmosphere at a tiny 0.0001%, it’s pretty expensive. Where does it come from?Īt least the billionaire Luthor would be able to afford a canister of Krypton. If Lex Luthor sprayed a bunch of real Krypton gas at his enemy, Superman would probably just blink and raise an eyebrow. It’s hardly going to cause you problems, let alone bring Superman to his knees. That means it’s one of the most non-reactive, inert substances on the planet. Krypton’s a gas, and not just any sort of gas, it’s a noble gas. The thing is, Krypton in real life isn’t anywhere near as exciting. In the comics, Kryptonite is a green, glowing material that is said to be the only thing that can defeat Superman. Natural krypton is a mixture of six stable isotopes. 
The boiling point of krypton is -243.81 F (-153.23 C), and its freezing point is only slightly lower at -251.27 F (-157.37 C). To coincide with our Superhero School Holidays, we’re celebrating a noble gas and the home of Superman Krypton. At extremely low temperatures, krypton may exist as a liquid or a solid. As you know at Science Space Wollongong, this year we’re featuring one element a month. Our next element featured as part of the International Year of the Periodic Table is Krypton. Look! It’s a bird! It’s a plane! No! It’s a largely nonreactive noble gas that fluoresces under certain conditions…
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
